First Pharmacologic Approach to Target Pinguecula’s Root Cause
Cloudbreak Pharma is pioneering the first FDA-approvable pharmaceutical treatment for pinguecula — a condition affecting over 1 billion people worldwide with no approved pharmacological options. Our lead program, CBT-004, is a proprietary Multi-Kinase Inhibitor (MKI) eye drop designed to simultaneously target the multiple signaling pathways that drive the abnormal fibrovascular changes of pinguecula, rather than only targeting symptoms.
Multi-Kinase Inhibitor Platform
TARGETING THE FULL DISEASE CASCADE, NOT JUST ONE PATHWAY
Current management of pinguecula is aimed at easing symptoms and controlling surface inflammation rather than eliminating the underlying degenerative change. Lubricating drops, anti-inflammatory medications, and environmental modifications can reduce irritation, but relief is often incomplete and temporary, and the visible lesion typically remains. In more bothersome cases, patients may still struggle with redness, discomfort, or contact lens intolerance, and only a very limited subset of patients are offered surgery for persistent inflammation or cosmetic concern, underscoring the need for better, targeted therapies.
How Pinguecula Develops
Chronic UV radiation exposure initiates a cascade of cellular damage in the conjunctival tissue:
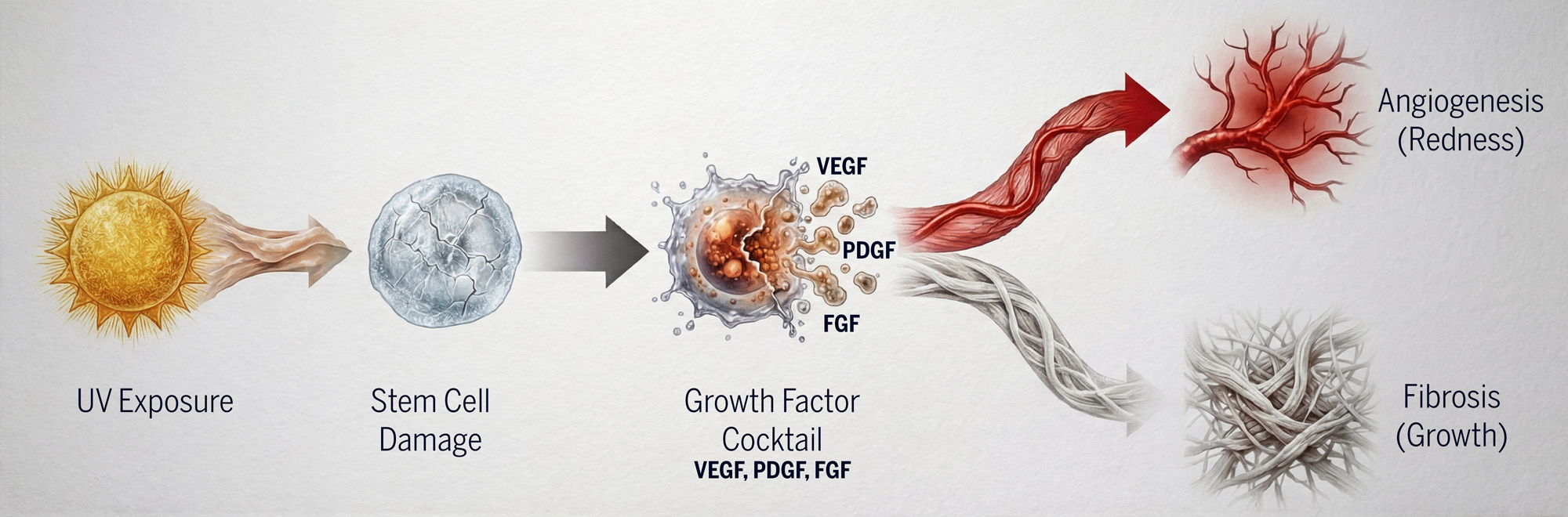
UV Exposure: DNA damage to conjunctival cells and limbal fibroblasts
Cell Activation: Upregulation of inflammatory cytokines (TNF-α, IL-6, IL-8)
Growth Factor Release: Overexpression of VEGF, PDGF, FGF, TGF-β
Vascularization & Fibrosis: Abnormal fibrovascular deposit formation
How CBT-004 Intervenes
Because pinguecula involves multiple overlapping signaling pathways, single-target therapies are insufficient. When one pathway is blocked, others compensate — allowing disease to persist or progress. Cloudbreak’s MKI platform prevents this through simultaneous multi-pathway inhibition.
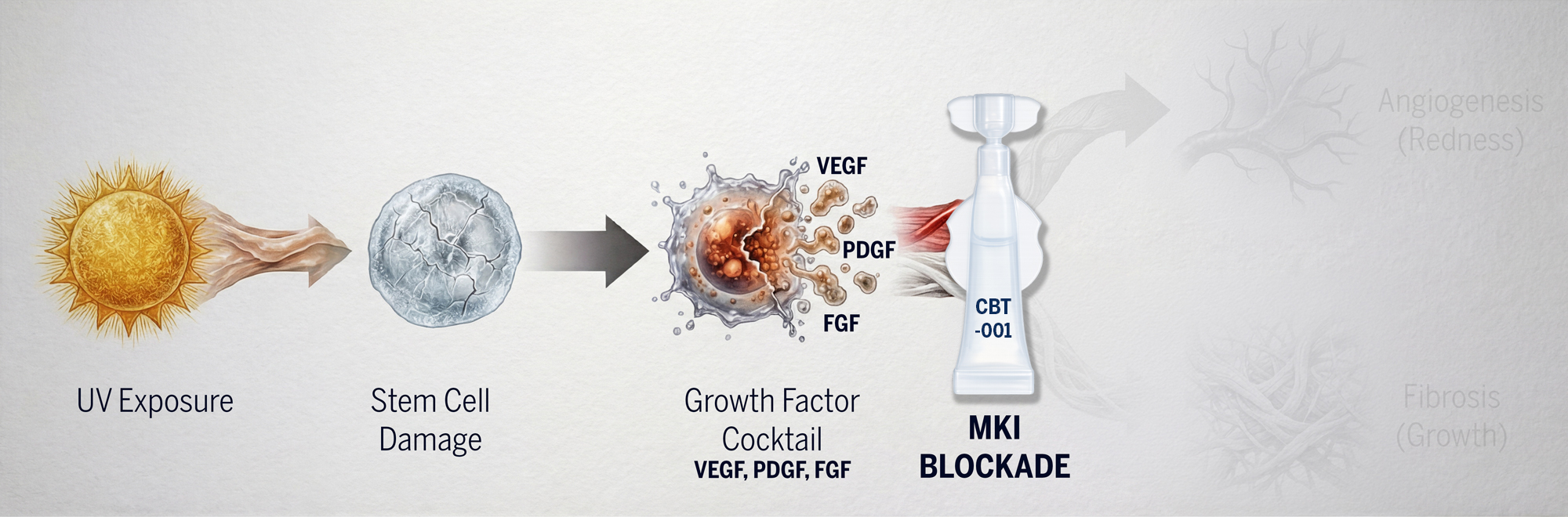
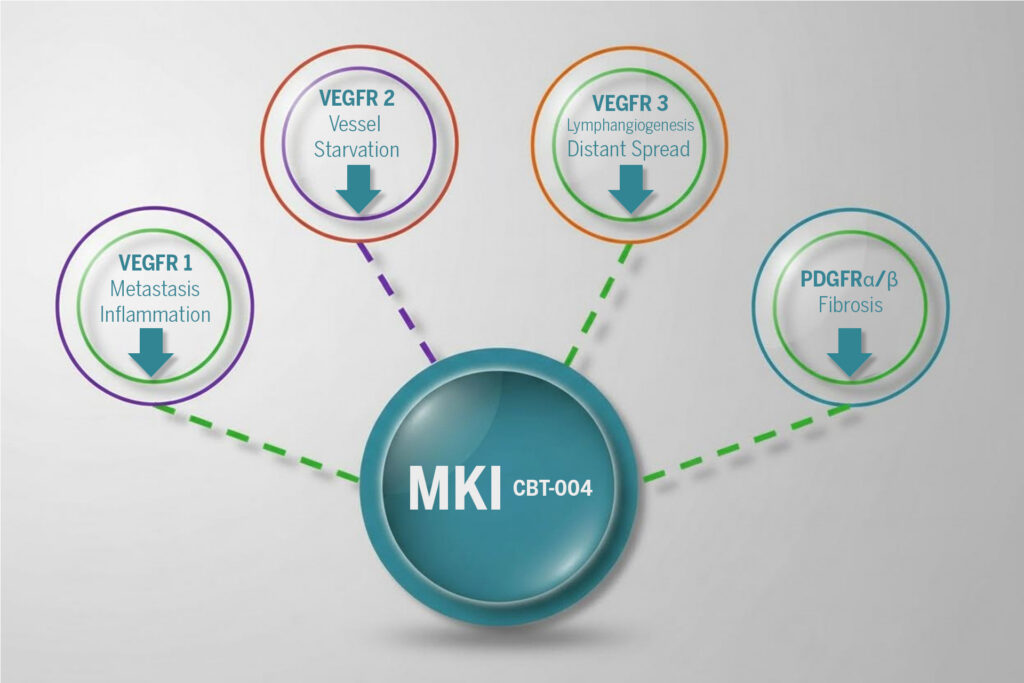
CBT-004 targets:
VEGFRs: Inhibits pathological angiogenesis/vascularization
PDGFRs: Reduces fibroblast activation and connective tissue remodeling
FGFRs: Suppresses further inflammatory cell signaling
CBT-004: Clinical Proof of Concept
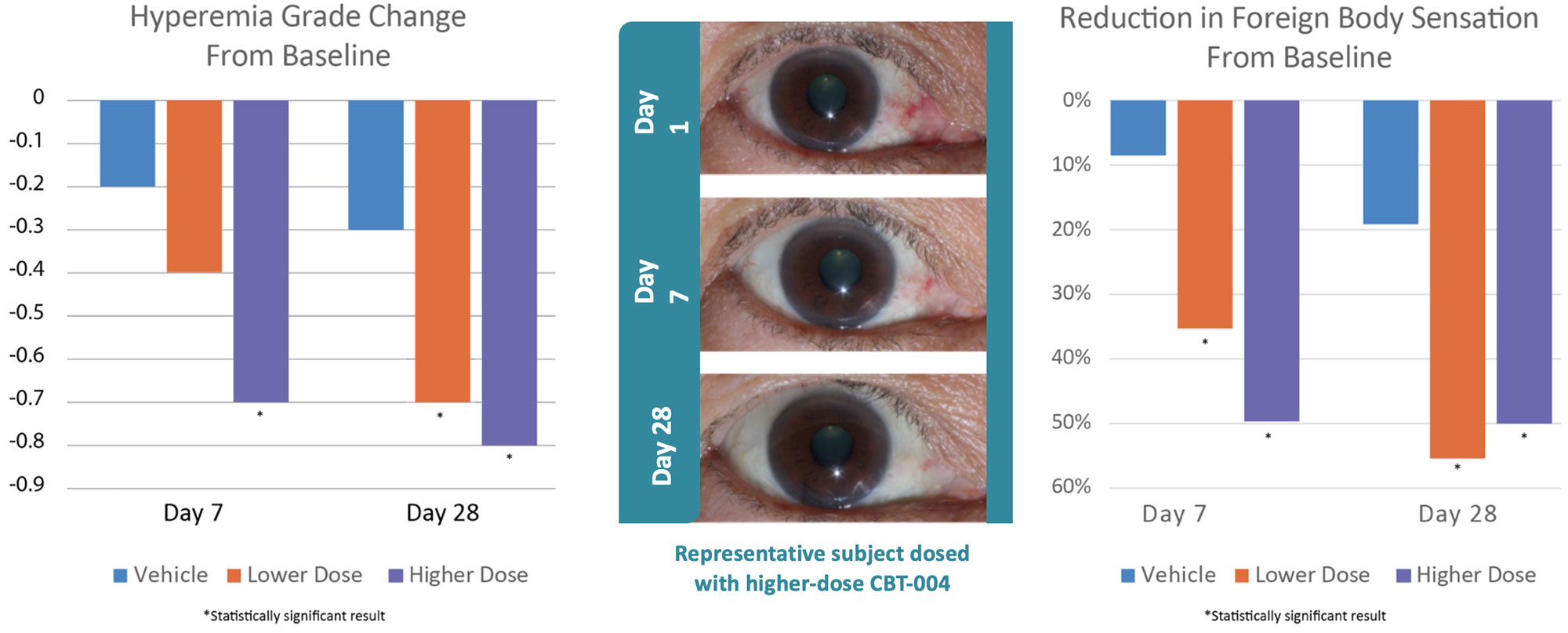
Phase 2 Results: Primary Endpoint Achieved.
First Pharmacologic Proof of Disease Modification in Pinguecula.
| Treatment Group | Hyperemia Grade Change | p-value vs Vehicle | |
|---|---|---|---|
| Vehicle Control | 0.30 | --- | N/A |
| CBT-004 0.05% | -0.68 | p = 0.043 | Statistically significant |
| CBT-004 0.075% | -0.78 | p = 0.012 | Statistically significant |
KEY CLINICAL HIGHLIGHTS

DOSE RESPONSE
Clear relationship observed between dose and efficacy (0.075% > 0.05% > vehicle) for hyperemia grade change

EARLY ONSET
High dose showed a significant effect as early as Day 7
(p = 0.007)

CLEAR EFFICACY
Both doses superior to vehicle and are superior to current SOC options

WELL TOLERATED
No systemic effects; mild & transient local AEs only
The time course data illustrates that CBT-004’s effects emerge rapidly and are sustained through the primary endpoint. The 0.075% high-dose arm demonstrated a statistically significant separation from vehicle as early as Day 7 (p = 0.007) — a clinical indicator of rapid onset of pharmacological activity. Both doses continued to show significant benefit at Day 28, the primary endpoint. This early and sustained efficacy profile supports CBT-004’s potential as the first meaningful pharmacological therapy for vascularized pinguecula.
PATIENT-REPORTED SYMPTOM: FOREIGN BODY SENSATION
Primary Complaint from People with Pinguecula
Why Measuring Both Hyperemia and Foreign Body Sensation (FBS) Matters Clinically:
A treatment that reduces observable vascular pathology (hyperemia) without improving how patients feel (FBS and related symptoms) would offer limited real-world therapeutic value. Conversely, a treatment that only masks symptoms without addressing underlying disease mechanisms would not represent true disease modification.
CBT-004’s dual impact — on both objective vascular signs and subjective symptom burden — validates its potential as a comprehensive, disease-modifying therapy that addresses the full spectrum of pinguecula pathology.
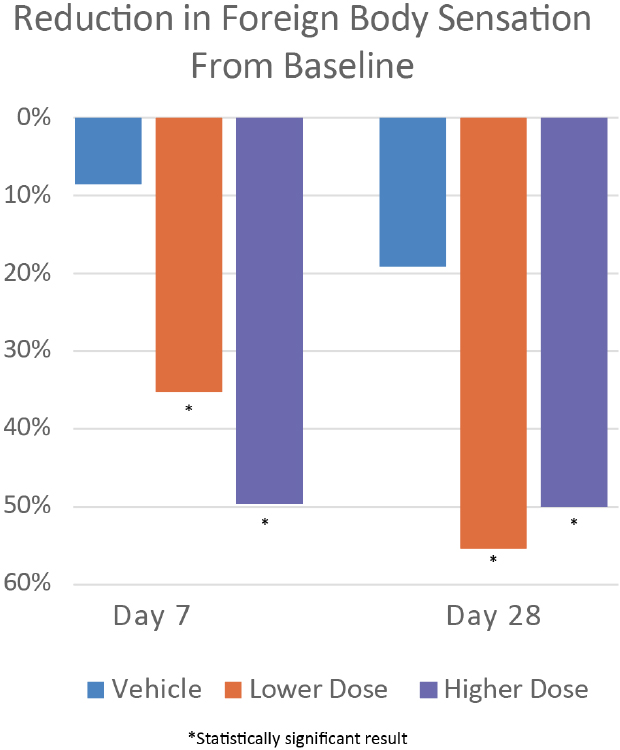
CBT-004 Phase 2 Results: Foreign Body Sensation (VAS Score, 0-100 Scale)
What Was Measured: Foreign body sensation was assessed using a Visual Analog Scale (VAS) where patients rated their current level of grittiness/discomfort on a 0-100 scale (0 = no discomfort; 100 = worst imaginable discomfort).
Key Clinical Findings:
- Rapid Onset of Symptom Relief: Both CBT-004 doses showed statistically significant FBS improvement as early as Day 7 — within just one week of treatment initiation
- Sustained Efficacy at Primary Endpoint: At Day 28, both doses demonstrated clinically meaningful and statistically significant FBS reduction compared to vehicle
- 0.05% dose: -23.11 points (p = 0.006) — represents ~56% improvement from baseline
- 0.075% dose: -18.76 points (p = 0.045) — represents ~51% improvement from baseline
- Persistent Benefit During Follow-Up: Even at Day 56 (4 weeks after treatment cessation), both doses maintained statistically significant symptom improvement, suggesting potential durability of effect
- Consistency Across All Time Points: FBS showed significant improvement at every measured time point (Days 7, 28, and 56) for both doses — one of the most consistent secondary endpoints in the study
Addressing a Critical Unmet Need
More Than a Billion Patients Waiting for a Real Treatment

1+ Billion Affected
Over 1 billion people globally
are affected by pinguecula
No Treatments
No FDA-approved drug therapies exist today

Phase 2 Complete
CBT-004 has achieved first-in-class
proof-of-concept
Despite its enormous global prevalence, pinguecula has been historically neglected in ophthalmic drug development.
The condition sits in a therapeutic gap, considered by many providers as “not serious enough” for intervention, yet causing real daily symptoms that affect patients’ comfort, vision quality, and quality of life. Eye care professionals currently lack any evidence-based, disease-modifying pharmacological tool to offer their patients.
Cloudbreak’s CBT-004 changes that. For the first time, a clinically validated approach that directly targets the vascular and fibrotic changes driving pinguecula could be offered with a well-tolerated topical formulation that fits seamlessly into existing eye care workflows.
One Platform,
Multiple Conditions
The same multi-kinase inhibitor mechanism that enables CBT-004’s activity in pinguecula positions Cloudbreak’s platform for broader application across anterior segment diseases characterized by abnormal fibrovascular activity. By targeting shared upstream signaling pathways — VEGF, PDGF, FGF, and TGF-β — the platform has potential utility in corneal neovascularization and other conditions where vascular and fibrotic dysregulation drives disease.
Why This Matters to People with Pinguecula,
Professionals & Investors
Pinguecula is a chronic, symptomatic ocular surface disease that can affect comfort, appearance, and daily functioning for millions of adults, it is more than a cosmetic concern. Framing pinguecula as a true disease state, rather than an incidental finding, highlights its impact on patients, the demands it places on clinical practice, and the broader burden on healthcare systems and payers.
For patients, this means ongoing irritation, redness, and lifestyle limitations, often without a durable treatment option. For eye care professionals, it represents a frequent, recurring reason for visits that is managed largely with short-term symptomatic measures. For investors, the large, underserved patient population and lack of disease‑modifying therapies point to a meaningful opportunity for innovation.
At Cloudbreak Pharma, we are advancing late‑stage drug candidates specifically designed to intervene earlier in this disease, with the goal of reducing symptoms, stabilizing the ocular surface, and ultimately decreasing the need for procedural or surgical interventions over time.
People with Pinguecula
Find out more about the causes of pinguecula and current management options
Professionals
Find out how Multi-Kinase Inhibitors may represent the first disease modifying approach to treating pterygium
Investors
Find out more about Cloudbreak Pharma as a potential investment opportunity on the Hong Kong Stock Exchange (2592.HK)