First Pharmacologic Approach to Target Pterygium’s Root Cause
Cloudbreak Pharma is pioneering the first FDA-approvable pharmaceutical treatment for pterygium — a condition affecting over 15 million people in the U.S. with no approved pharmacological options. Our lead program, CBT-001, is a proprietary Multi-Kinase Inhibitor (MKI) eye drop designed to simultaneously target the multiple signaling pathways that drive the abnormal fibrovascular changes of pterygium, rather than simply masking symptoms.
MULTI-KINASE INHIBITOR PLATFORM
TARGETING THE FULL DISEASE CASCADE, NOT JUST ONE PATHWAY
Current management of pterygium is aimed at easing symptoms and controlling surface inflammation rather than eliminating the underlying degenerative change. Lubricating drops, anti-inflammatory medications, and environmental modifications can reduce irritation, but relief is often incomplete and temporary, and the visible lesion typically remains. In more bothersome cases, patients may still struggle with redness, discomfort, or contact lens intolerance, and only a small minority are offered surgery for persistent inflammation or concerns about appearance, underscoring the need for better, targeted therapies.
How Pterygium Develops
Chronic UV radiation exposure initiates a cascade of cellular damage in the conjunctival tissue:
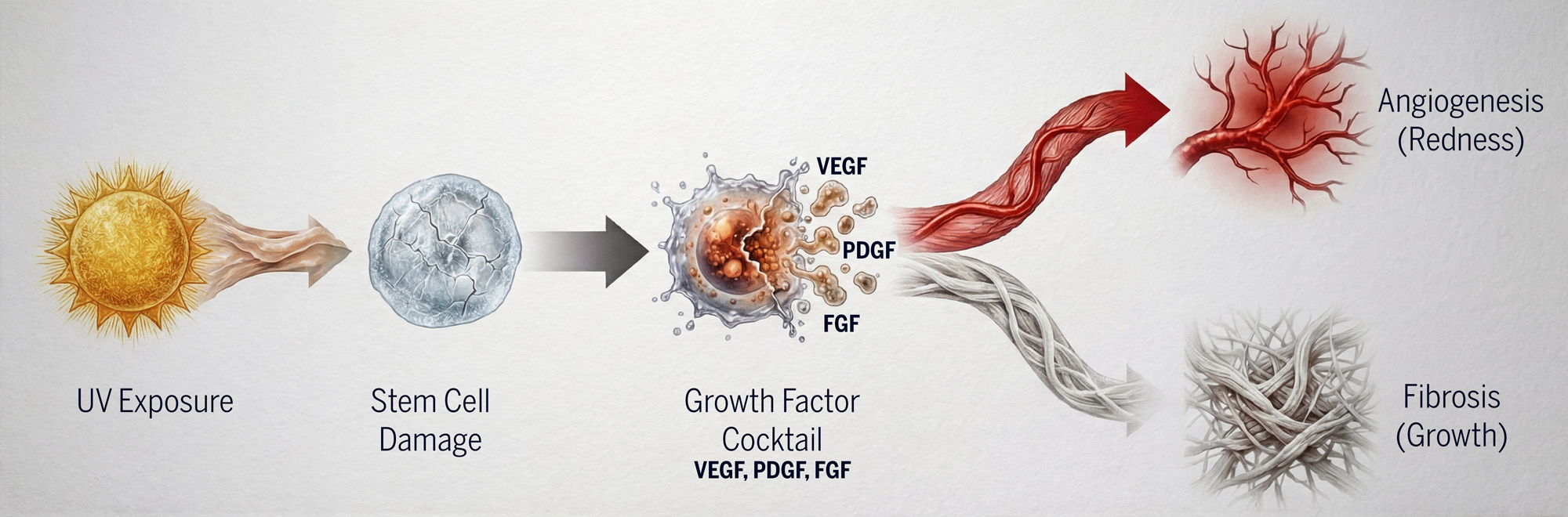
UV Exposure: DNA damage to conjunctival cells and limbal fibroblasts
Cell Activation: Upregulation of inflammatory cytokines (TNF-α, IL-6, IL-8)
Growth Factor Release: Overexpression of VEGF, PDGF, FGF, TGF-β
Vascularization & Fibrosis: Abnormal fibrovascular deposit formation
How CBT-001 Intervenes
Pterygium’s overlapping signaling pathways render single-target therapies ineffective — compensatory mechanisms allow disease to persist. Cloudbreak’s MKI platform overcomes this through simultaneous multi-pathway inhibition.“
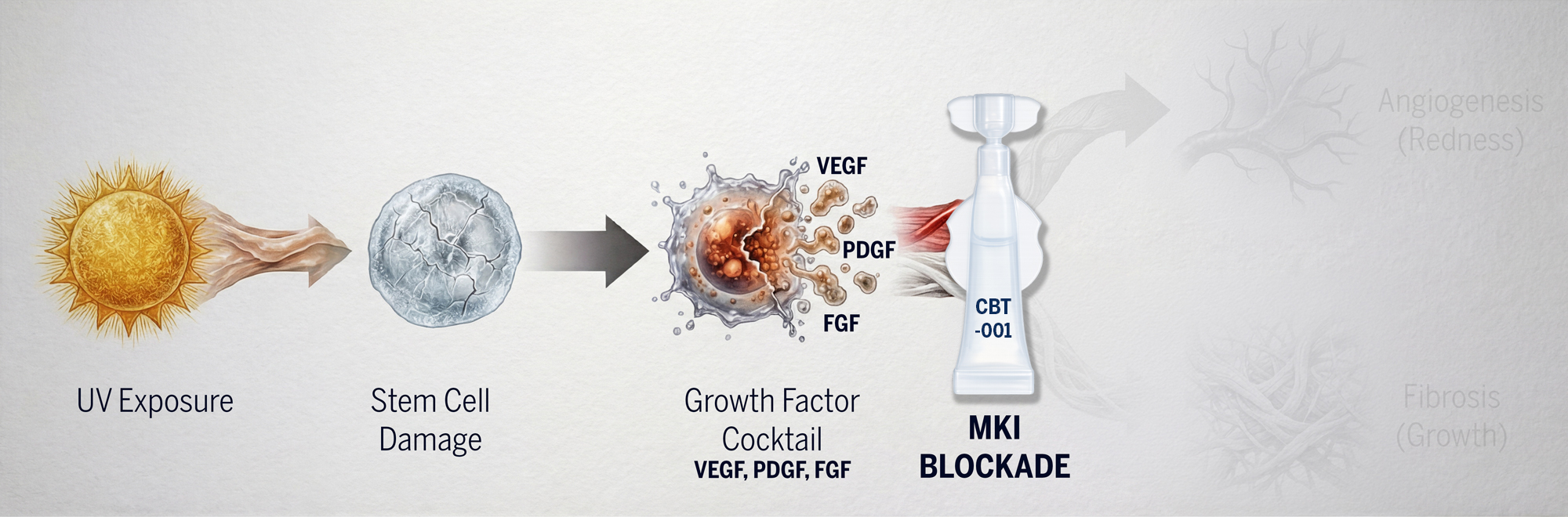
CBT-001 targets:
VEGFRs: Inhibits pathological angiogenesis/vascularization
PDGFRs: Reduces fibroblast activation and connective tissue remodeling
FGFRs: Suppresses further inflammatory cell signaling
CBT-001: CLINICAL PROOF OF CONCEPT
Phase 2 Results: Dual Evidence of Disease Modification
Objective Vascular Signs AND Patient Symptoms Improved
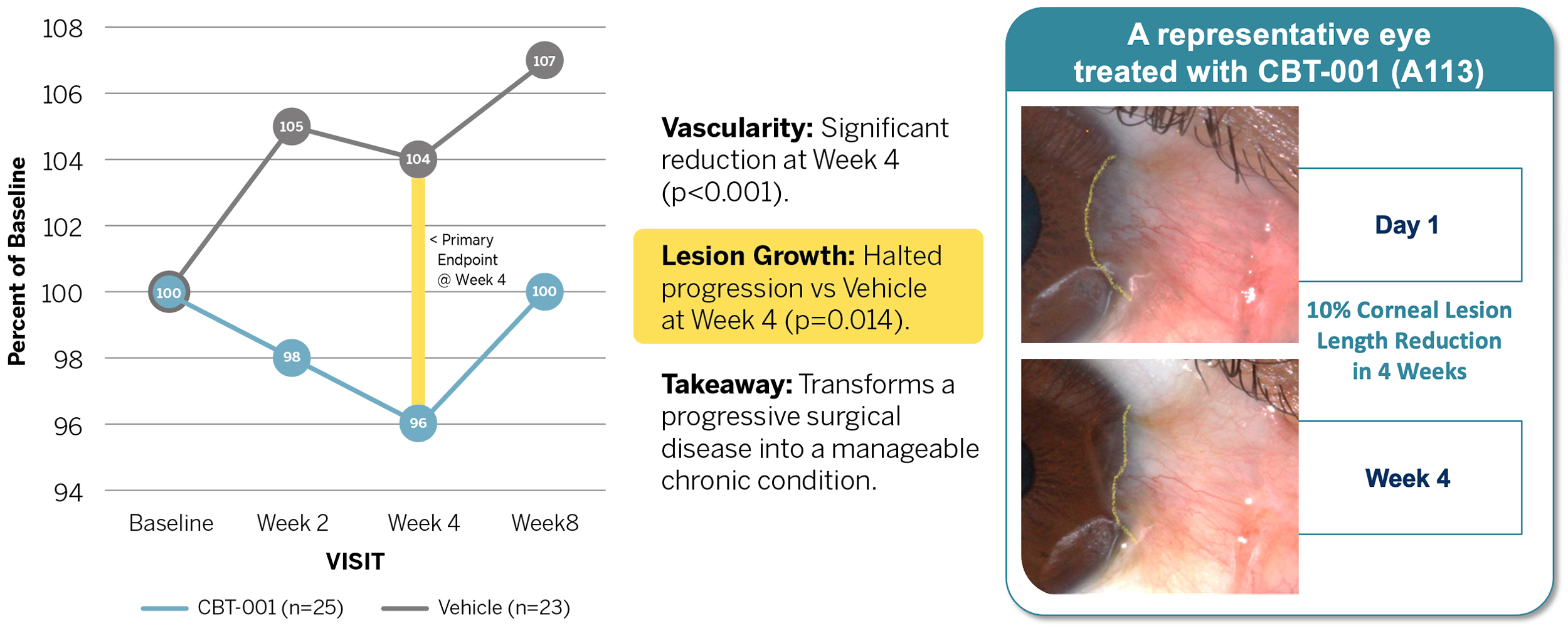
KEY CLINICAL HIGHLIGHTS

VASCULARITY REDUCTION
Mean −0.8 grade reduction at Week 4 vs. no change for vehicle; treatment difference of −0.76 grade (p<0.001)

LESION GROWTH HALTED
3.3% reduction in lesion length vs. 8.0% increase in vehicle at Week 4 (p=0.006)

WELL TOLERATED
Minimal systemic exposure (>3,400× lower than oral nintedanib); mild & transient local AEs only
Phase 2 Results: Dual Evidence of Disease Modification
Objective Vascular Signs AND Patient Symptoms Improved
PRIMARY ENDPOINT ACHIEVED
CBT-001 0.2% demonstrated highly statistically significant reduction in pterygium vascularity (objective vascular measure) vs. vehicle at Week 4 (p < 0.001)
PATIENT SYMPTOMS SIGNIFICANTLY IMPROVED
Ocular symptoms (including red eye, irritation, and discomfort) showed meaningful improvement across multiple PSLQ domains, with red eye symptom achieving statistical significance (p = 0.038) at Week 4
PRIMARY ENDPOINT: PTERYGIUM VASCULARITY
Objective Vascular Pathology
What Was Measured: Pterygium vascularity — the characteristic “fleshy” fibrovascular tissue with prominent vessel network and vascular turgescence — was graded on a validated 5-point scale (0-4) by an independent reading center (Doheny Eye Institute) using standardized digital photography. As noted in the CSR:
“Pterygium vascularity grading assesses the vascular turgescence related to the severity of the fibrovascular proliferation, as highlighted by the vessel network morphology. This is of major interest because it drives the visual prognosis.“
Week 4 Primary Endpoint Results — Pterygium Vascularity Grade Change from Baseline:
| Treatment Group | Baseline Mean | Mean Change from Baseline | Within-Group p-value | Treatment Difference vs Vehicle | Between-Group p-value |
|---|---|---|---|---|---|
| Vehicle Control | 3.0 ± 0.8 | 0.0 ± 0.5 | p > 0.999 (NS) | ––– | ––– |
| CBT-001 0.2% | 2.9 ± 0.7 | -0.8 ± 0.7 | p < 0.001 | -0.76 grade | p < 0.001 |
Note: Negative values indicate reduction in vascularity grade (improvement). Higher magnitude of reduction = greater efficacy.
Key Clinical Findings:
- Highly Significant Primary Endpoint: At Week 4 (primary endpoint), CBT-001 0.2% demonstrated a mean vascularity reduction of -0.8 grade compared to no change (0.0) in the vehicle group
- Treatment difference: -0.76 grade (p < 0.001)
- This represents nearly a one full grade improvement in vascularity severity
- Rapid Onset: Significant separation from vehicle was evident as early as Week 2 (-0.6 vs +0.1, p < 0.001)
- Sustained Efficacy: Statistically significant treatment differences persisted through Week 16 (p ≤ 0.008)
- Clinically Meaningful Responder Rates:
- 68% of CBT-001 patients showed ≥1 grade improvement at Week 4 vs only 13% of vehicle patients (p < 0.001)
- 60% responder rate as early as Week 2 vs 0% in vehicle (p < 0.001)
CBT-001 0.2% Ophthalmic Solution — Pterygium Vascularity Outcomes
Phase 2 Randomized Controlled Trial · Primary Endpoint: Week 4 · Vascularity Score (0–4 scale)

PATIENT-REPORTED SYMPTOMS: PSLQ QUESTIONNAIRE
(Subjective Quality of Life)
Why Patient Symptoms Are Critical:
Pterygium is not just an appearance issue — it causes significant functional impairment and symptom burden. As documented in the CBT-001 CSR Introduction:
“Although pterygium is non-malignant, it can cause significant ocular symptoms, including hyperemia, irritation, pain, and foreign body sensation, thus it impacts the quality of patients’ life.“ (Citing Jaros and DeLuise, 1988)
Beyond the visible vascular pathology that clinicians observe and grade, patients experience daily discomfort, visual disturbances, and quality-of-life impacts that drive treatment-seeking behavior and determine real-world therapeutic value.
The PSLQ (Pterygium Symptom and Life Quality) Questionnaire:
The PSLQ is a validated 15-question instrument derived from the Visual Functioning Questionnaire-25 (VFQ-25) and Ocular Surface Disease Index (OSDI). It assesses three critical domains:
- Ocular Symptoms (6 questions): Frequency of eye redness, irritation, discomfort, foreign body sensation, and related symptoms
- Vision-Related Functioning (4 questions): Impact on daily visual tasks and activities
- Quality of Life Impact (5 questions): How pterygium affects overall quality of life, social interactions, and daily functioning
Patients rated each symptom on a 5-point frequency scale (0 = none of the time; 4 = all the time), with lower scores indicating better outcomes.
CBT-001 Phase 2 Results: PSLQ Outcomes
Overall PSLQ Score: While between-group differences did not reach statistical significance for overall PSLQ score, both CBT-001 and vehicle groups showed improvements from baseline, suggesting potential trial design effects (e.g., placebo response, natural variability). However, CBT-001 demonstrated:
- Statistically significant within-group improvements at Weeks 2, 4, 8, 16, and 24 (p ≤ 0.024)
- Sustained benefit across all follow-up timepoints through 24 weeks
- Numerically greater improvements compared to vehicle at most timepoints
PSLQ Ocular Symptom Subscore:
The Ocular Symptom domain — which captures the most direct symptom burden from pterygium (redness, irritation, discomfort, grittiness) — showed the same pattern:
- CBT-001: Statistically significant improvements from baseline at Weeks 2, 4, 8, 16, 24 (p ≤ 0.024)
- Vehicle: Improvements only at Weeks 2, 4, 8 (p ≤ 0.035), with effects diminishing by Week 16
- Trend toward greater benefit with CBT-001 treatment despite not achieving between-group statistical significance
PSLQ Quality of Life Impact:
At Week 8, CBT-001 showed statistically significantly greater improvement in Quality of Life Impact compared to vehicle (p < 0.05), validating that vascular improvements translate into meaningful patient-centered outcomes.
PSLQ Red Eye Symptom — SIGNIFICANT RESULT AT WEEK 4
Subset Analysis: Patients with Baseline Red Eye Score >1 (Moderate-to-Severe Red Eye)
A clinically important subset analysis focused on patients who reported frequent red eye at baseline (score >1, indicating red eye “half the time” to “all the time”). This subset represented patients with the most bothersome and visible symptom.
Red Eye Symptom Results at Week 4 (Primary Endpoint):
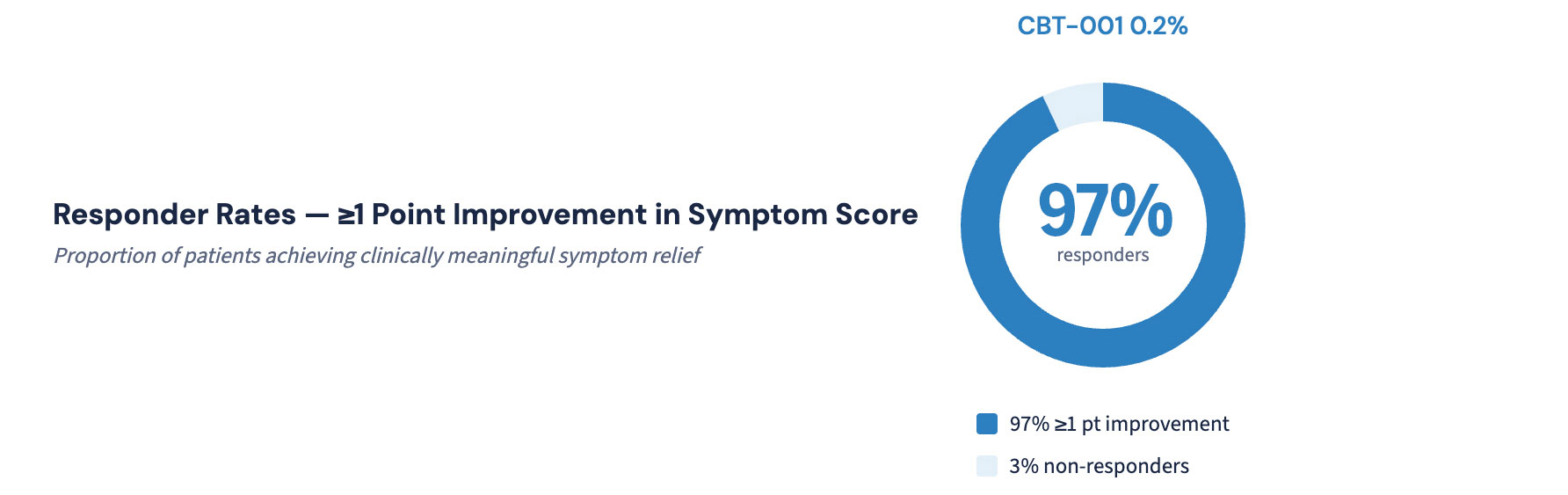
| Treatment Group | Patients with ≥1 Point Improvement | Mean Red Eye Score Reduction |
|---|---|---|
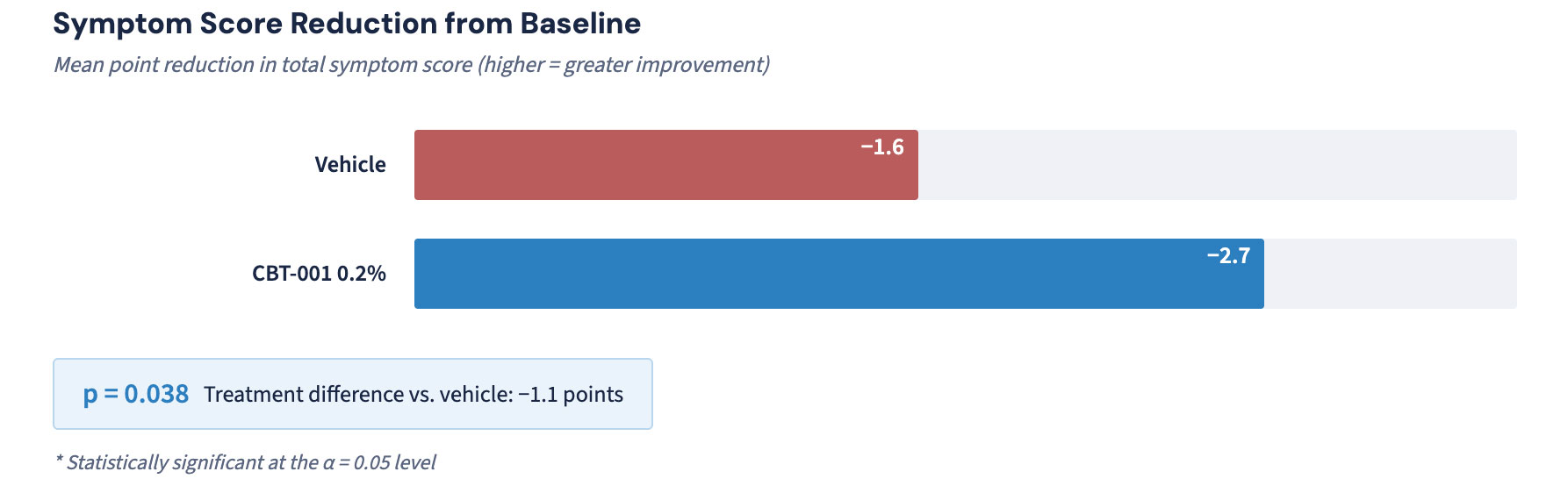
| Vehicle Control | 7/11 (64%) | -1.6 points |
| CBT-001 0.2% | 13/14 (97%) | -2.7 points |
Treatment Difference: -1.1 points (p = 0.038)
Key Clinical Significance:
- 93% responder rate in CBT-001 group vs 64% in vehicle
- Nearly double the symptom improvement (-2.7 vs -1.6 points)
- Statistically significant despite small sample size, demonstrating robust effect
- Red eye is the most visible, socially impactful symptom for pterygium patients — improvement in this domain addresses both physical discomfort and cosmetic concerns
Red eye — the most visibly apparent and socially impactful symptom of pterygium — showed statistically significant improvement with CBT-001 treatment. This dual impact on both vascular pathology (vascularity grade) and symptom burden (red eye, ocular discomfort) validates CBT-001’s disease-modifying potential across the full spectrum of pterygium manifestations.
KEY CLINICAL HIGHLIGHTS
| Vascularity | Symptoms | Responder Rate | Rapid Onset | Well-tolerated |
|---|---|---|---|---|
| Highly significant reduction in separation in pterygium vascularity at Week 4 • -0.8 grade vs 0.0 • p < 0.001 | Red eye symptom (most visible, bothersome symptom) showed significant improvement (93% responder rate) | 68% of patients showed >1 grade vascularity improvement at Week 4 vs only 13% vehicle (p<0.001) | Significant separation from vehicle as early as Week 2 (p<0.001), with sustained benefit through Week 16 | No systemic safety concerns; mild and transient local AEs only |
CLINICAL SIGNIFICANCE STATEMENT
DUAL EVIDENCE OF DISEASE MODIFICATION
CBT-001 is the first pharmacologic treatment to demonstrate disease modification in pterygium across BOTH:
OBJECTIVE VASCULAR PATHOLOGY (Pterygium Vascularity)
✓ Highly significant vascularity reduction (-0.8 grade, p<0.001 at Week 4)
✓ Independent reading center grading ensures unbiased assessment
✓ 68% responder rate (≥1 grade improvement) vs 13% vehicle
✓ Addresses the visible fibrovascular proliferation that drives visual threat
PATIENT-REPORTED SYMPTOM BURDEN (PSLQ Outcomes)
✓ Red eye symptom significantly improved (p=0.038, 93% responder rate)
✓ Ocular symptoms showed sustained improvements through 24 weeks
✓ Quality of Life Impact significantly better than vehicle at Week 8
✓ Validates real-world benefit addressing both discomfort and cosmetic concern
This dual impact demonstrates that CBT-001’s multi-kinase inhibitor platform addresses root pathophysiology — not just cosmetic appearance — delivering meaningful clinical benefit across the disease spectrum: vascular pathology, symptom burden, quality of life, and visual prognosis.
ADDRESSING CRITICAL UNMET NEED
The Problem Over 400 million people worldwide suffer from pterygium, yet no FDA-approved pharmacological treatment exists. Current options — topical steroids, NSAIDs, and surgical excision — offer only temporary relief or mechanical removal, leaving the underlying vascular and fibrotic drivers of the disease unaddressed.
The Solution CBT-001 (nintedanib) is the first topical therapy to simultaneously target VEGFR, PDGFR, and FGFR — the core pathways driving pterygium’s progression. The clinical data presented here demonstrate its ability to reduce vascularity, inhibit lesion growth, and address the root cause of a condition that has gone untreated for far too long.
Why This Matters to People with Pterygium,
Professionals and Investors
People with Pterygium
Find out more about the causes of pterygium and current management options
Professionals
Find out how Multi-Kinase Inhibitors may represent the first disease modifying approach to treating pterygium
Investors
Find out more about Cloudbreak Pharma as a potential investment opportunity on the Hong Kong Stock Exchange (2592.HK)