A Novel Multi-Kinase Inhibitor Platform
Targeting the Root Cause,
Not Just Symptoms
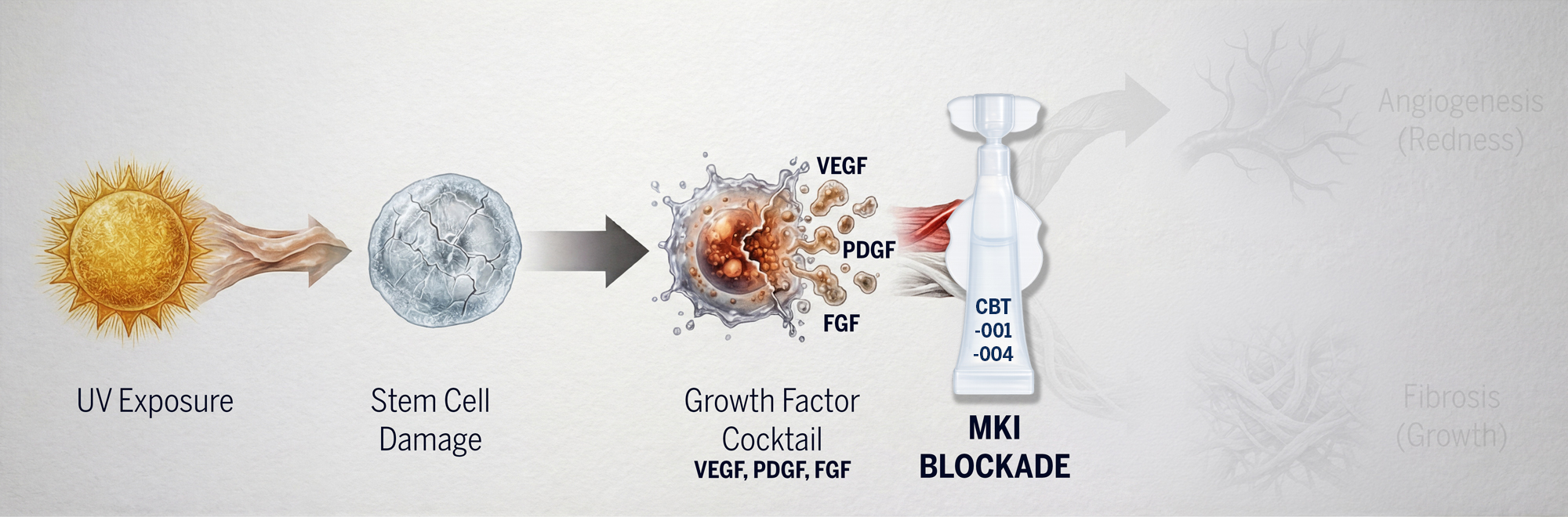
Cloudbreak Pharma is developing the first pharmacological treatments that address the underlying pathophysiology of pterygium and pinguecula. Our proprietary multi-kinase inhibitor (MKI) platform simultaneously targets multiple receptor tyrosine kinases that drive the abnormal fibrovascular growth characteristic of these conditions.
Unlike current treatments that only provide temporary symptom relief, Cloudbreak’s approach inhibits the inflammatory cytokines and growth factors upregulated by chronic UV exposure—directly addressing disease progression at its source.
Mechanism of Disease Modification
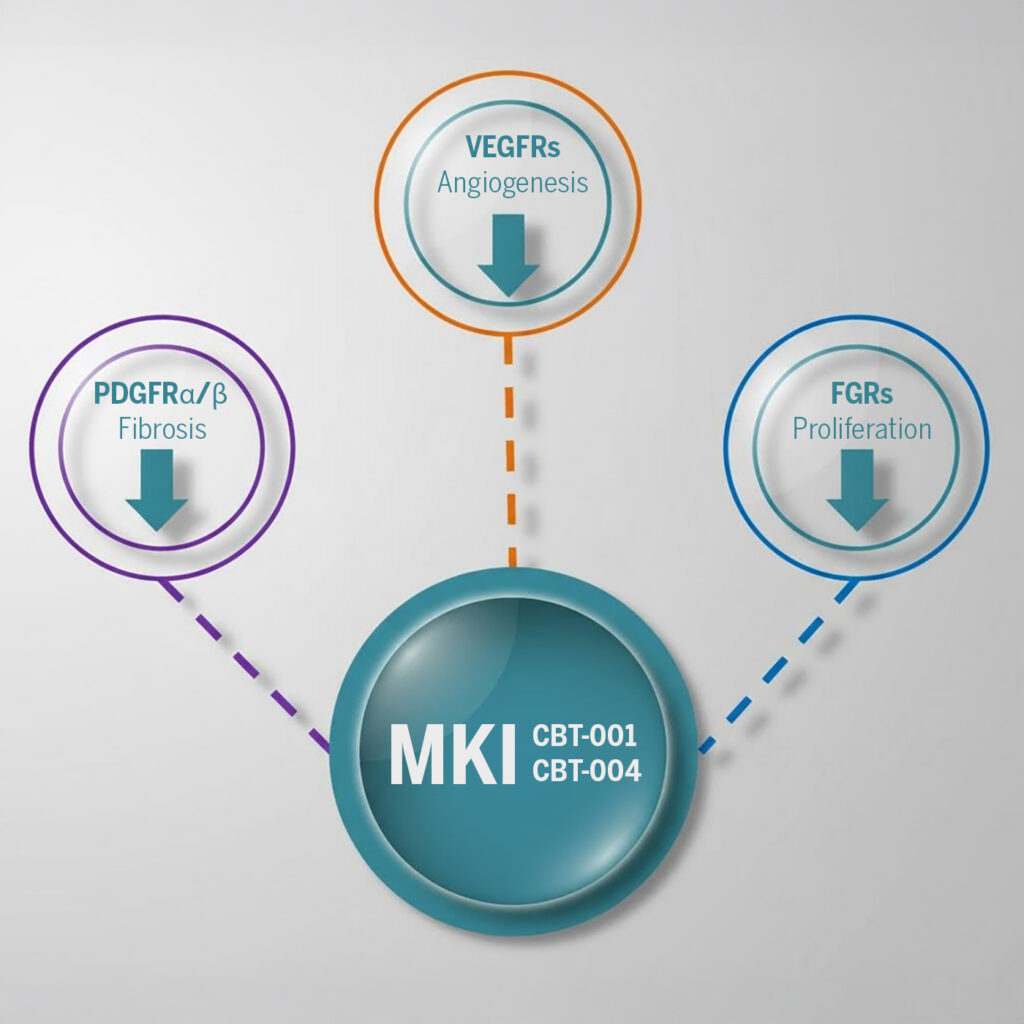
Multi-Target Intervention for Complete Disease Control
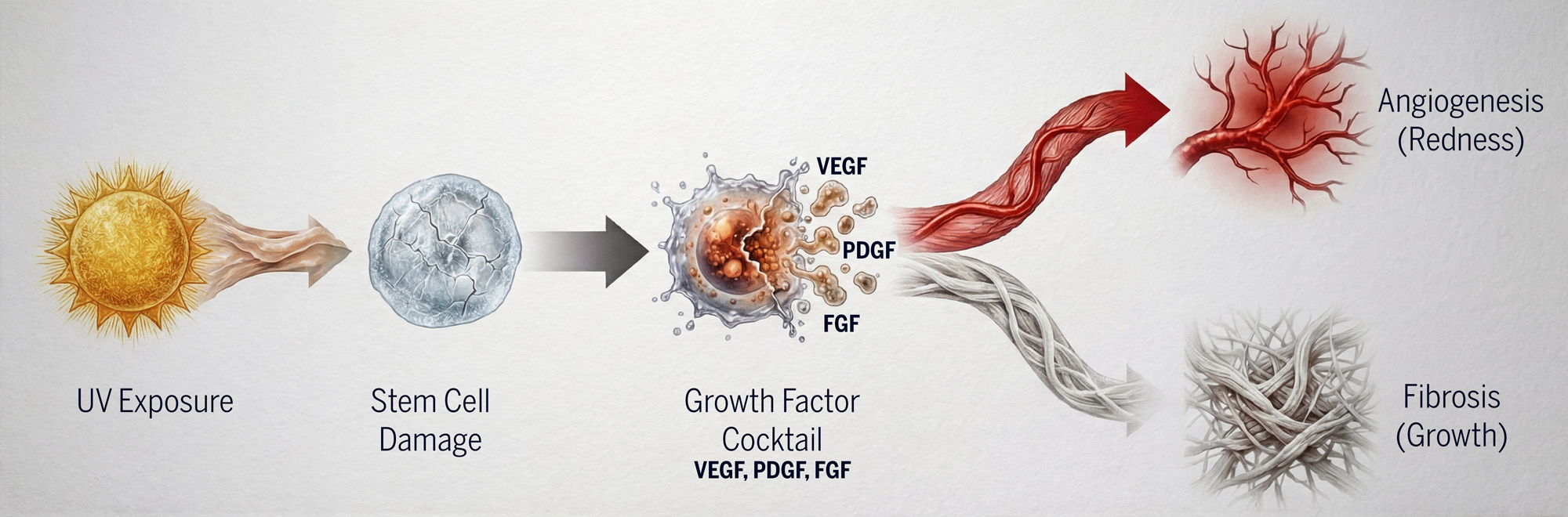
Pterygium and pinguecula result from a complex cascade triggered by chronic UV radiation exposure:
- UV damage to limbal stem cells and fibroblasts
- Activation of inflammatory and angiogenic pathways
- Upregulation of multiple growth factors (VEGF, PDGF, FGF, TGF-β)
- Progression of fibrovascular growth onto the cornea
Why Multi-Kinase Inhibition?
- ✓ Blocking multiple pathways simultaneously
- ✓ Addressing both angiogenesis AND fibrosis
- ✓ Delivering clinically proven disease modification
Addressing a Critical Unmet Need:
We are developing potentially the first FDA-approved pharmaceutical treatments for patients with pterygium and pinguecula
Our Lead Programs

CBT-001 for Pterygium
- Status: In Phase 3
- Indication: Primary pterygium
- Key Results: Phase 2 data demonstrated:
- Statistically significant reduction in pterygium vascularity (-1.2 grade, p<0.001 at Week 4)
- Statistically significant reduction of lesion length vs. vehicle (p=0.014)
- Well-tolerated safety profile with no systemic effects

CBT-004 for Pinguecula
- Status: Phase 3-ready
- Indication: Signs and symptoms of pinguecula
- Key Results: Phase 2 data demonstrated:
- Primary endpoint achieved: Both doses significantly superior to vehicle at Day 28
- High dose: -0.78 grade reduction in conjunctival hyperemia vs. vehicle (p=0.012)
- Low dose: -0.68 grade reduction (p=0.043) vs. vehicle
- Early onset of action with high dose: -0.63 grade reduction (p=0.007) at Day 7
- Improvement in ocular symptoms: statistically significant improvement in foreign body sensation with high dose at Day 7 and both doses at Day 28, vs. vehicle.
LIMITED UTILITY OF CURRENT STANDARD OF CARE
Current treatments miss the mark. None address the underlying pathophysiology
Artificial tears
Symptomatic only
Corticosteroids
Safety concerns limit to only temporary treatment
Surgery for Pterygium
~38% recurrence, $11K+ cost

What Patients Want
- 94% of pterygium patients surveyed expressed moderate to extreme interest in a new eye drop that could address symptoms and postpone or eliminate the need for surgery
- 87% have concerns about surgery (recurrence, cost, recovery time, pain)
- Eye care professionals report that patients often wait years before agreeing to surgery
Data on file

The Cloudbreak Difference
Our multi-kinase inhibitor approach offers the potential to:
- Modify disease process
- Reduce or eliminate pterygium/pinguecula symptoms
- Delay or avoid the need for surgery
- Provide a well-tolerated, convenient topical treatment
Platform Potential
Beyond Pterygium: A Versatile Platform for Anterior Segment Diseases
The multi-kinase inhibitor platform’s ability to simultaneously target angiogenesis, fibrosis, and inflammation positions it for potential application across multiple ocular surface diseases characterized by abnormal vascular and fibrotic growth, including:- Corneal neovascularization
- Corneal scarring
- Other anterior segment proliferative conditions