CBT-001
A potential first-in-class treatment for patients with pterygium
CBT-011 is currently in a Phase 3 clinical trial (NCT05456425) as a disease-modifying eyedrop therapy for pterygium.
突破性创新
Our Core Product CBT-001 is a potential first-in-class drug therapy using a multi-kinase inhibitor targeting platelet-derived growth factor receptors (“PDGFRs”), fibroblast growth factor receptors (“FGFRs”), and vascular endothelial growth factor receptors (“VEGFRs”). This unique eye drop is being developed as a treatment for pterygium, including reducing the size of the lesion as well as common, bothersome symptoms.
To our knowledge, there is currently no approved drug therapy for the treatment of pterygium globally, and the current existing treatment option for pterygium is surgical excision. If approved, CBT-001 is expected to be the first drug therapy globally for the treatment of pterygium and related bothersome symptoms, and to potentially reduce or postpone the need for surgical excision.
有药物效性能与安全性
2期临床试验7
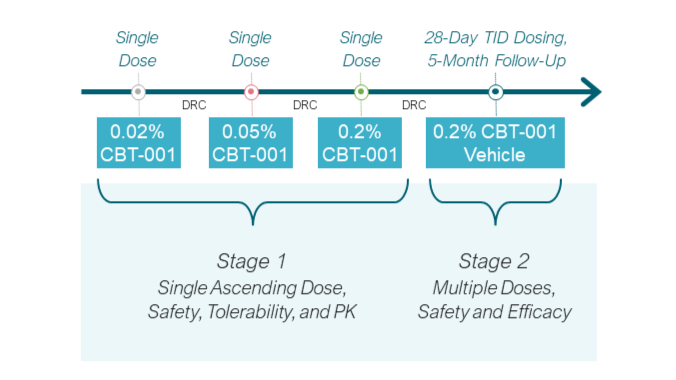
2期临床试验NCT03049852分为两个阶段。 第一阶段包括对 24 名患者进行 CBT-001 剂量递增,以确定最大耐受剂量。 结果证明了眼部安全性和耐受性,全身药物暴露可忽略不计。 在第 2 阶段,51 名患者接受了为期 4 周的 TID 周期 CBT-001 (n=25) 或载体 (n=23) 给药治疗,并随访至第 24 周。到第 4 周,用药组的平均血管分布评分比对照组显著下降, 并一直持续到第 16 周。此外,在第 4 周观察到病灶长度的平均变化比对照组明显减少,并一直持续到第 8周。7
CBT-001 具有良好的耐受性,CBT-001 组中常见报告的大多数不良事件是眼部不良事件,严重程度较轻,并在治疗后得到缓解。 没有患者因不良事件而停止 CBT-001 治疗。7
3期临床试验8
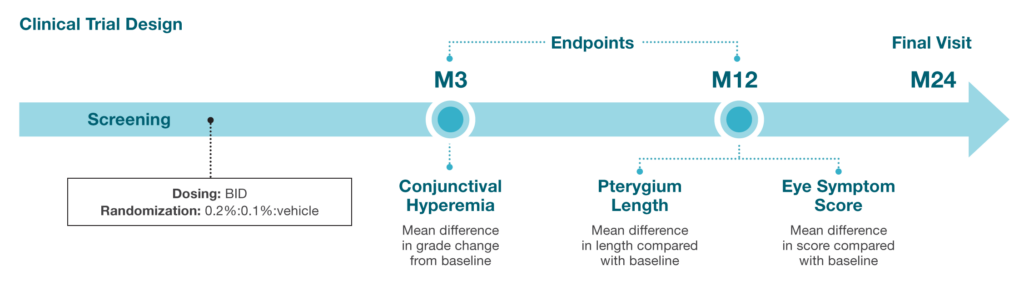
3期临床试验 (NCT05456425) is designed to evaluate the efficacy and safety of CBT-001 in patients with pterygium.8
方案标题:对翼状胬肉患者进行多中心、双盲、随机、载体对照 12 个月平行比较 0.1% 和 0.2% CBT-001 与载体(每天给药两次)的安全性和有效性
研究目的:评估 0.1% 和 0.2% CBT-001 乳剂每天两次给药,持续 24 个月,与载体相比,在减少翼状胬肉眼结膜充血和预防翼状胬肉进展方面的安全性和有效性
目标入组:600 名患者
主要纳入标准:
- 年龄≥12 岁的男性或女性
- 角膜上方翼状胬肉病变长度≥1.2毫米
- ≥3级结膜充血
主要排除标准:
- 过去 6 个月内进行过翼状胬肉切除手术
- 预期在研究入组一年内进行翼状胬肉手术
- 除翼状胬肉以外的具有临床意义的角膜异常
This Phase 3 clinical trial is fully enrolled with a data readout expected 3Q2026.
BID, twice daily; DRC, data review committee; FGFR, fibroblast growth factor receptor; M, month; PDGFR, platelet-derived growth factor receptor; PK, pharmacokinetics; TID, three times daily; VEGFR, vascular endothelial growth factor receptor.
1. Shahraki T, et al. Ther Adv Ophthalmol. 2021;13:25158414211020152. 2. Chu WK, et al. Eye (Lond). 2020;34(6):1047-1050. 3. Wanzeler ACV, et al. Clin Ophthalmol. 2018;12:833-837. 4. Palewski M, et al. Int J Environ Res Public Health. 2022;19:11357. 5. Hilberg F, et al. Cancer Res. 2008;68:4774–4782. 6. Yang R, et al. Invest Ophthalmol Vis Sci .2019;60(9):2087. 7. Whitcup SM, et al. Ophthalmol Sci. 2024. Publication in press. https://doi.org/10.1016/j.xops.2024.100502. 8. ClinicalTrials.gov. A clinical trial on safety and efficacy of CBT-001 in patients with pterygium. Updated March 15, 2024. Accessed March 15, 2024. https://clinicaltrials.gov/study/NCT05456425.